
Since entering the market in 2015, the CDK4/6 inhibitors―Pfizer’s Ibrance, Eli Lilly’s Verzenio/Verzenios and Novartis’ Kisqali―have become the standard-of-care for first-line advanced / metastatic HR-positive / HER2-negative breast cancer. While Ibrance falters in the pivotal PALLAS study, Verzenio succeeds in monarchE, paving the way for approval in the most lucrative subpopulation of breast cancer. Ibrance stumbles while Verzenio exceeds in early-stage HR-positive / HER2-negative breast cancer.While these results provide complimentary evidence for adding an immune checkpoint inhibitor to neoadjuvant chemotherapy for early-stage TNBC, differentiating between the two drugs is currently challenging EFS data could prove pivotal to the future roles of these drugs in the early-stage setting. Previously, data from KEYNOTE-522 at SABCS 2019 demonstrated that adding neoadjuvant Keytruda to chemotherapy in patients with lymph-node-positive early-stage TNBC significantly improved pCR in the ITT population (64.8% vs. 41.1%) 4, according to data presented at ESMO 2020 from IMpassion031. In high-risk early-stage TNBC, the addition of neoadjuvant Tecentriq to chemotherapy has shown a significant improvement in pCR rates regardless of PD-L1 status (57.6% vs. The reasons behind these conflicting data are currently unknown and under investigation but nonetheless, will likely influence physicians’ choice of immune checkpoint inhibitor. 5.6 months) in inoperable/metastatic PD-L1-positive TNBC (KEYNOTE-355) 3. 
In previously reported data, the addition of Merck & Co.’s Keytruda to several chemotherapy backbones (including Abraxane and paclitaxel) led to a statistically significant improvement in median PFS (9.7 months vs. However, in data presented for IMpassion131, no difference was found between metastatic TNBC patients that received Tecentriq plus paclitaxel and placebo plus paclitaxel in terms of PFS and OS, irrespective of PD-L1 status 2. Analysis from this trial presented at ESMO 2020 demonstrated a statistically significant median OS benefit for Tecentriq + Abraxane (25.4 months) compared to placebo + Abraxane (17.9 months) in the PD-L1-positive population 1. 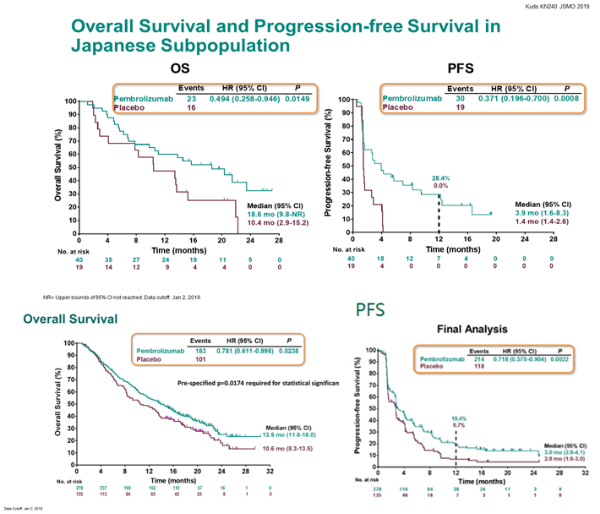
Roche’s Tecentriq was the first immune checkpoint inhibitor approved for metastatic PD-L1-positive triple-negative breast cancer (TNBC), in combination with Abraxane based on positive PFS data in IMpassion130. Mixed results for Tecentriq puts Keytruda in pole position in metastatic triple-negative breast cancer early-stage disease still up for grabs. Evolving landscape of immune checkpoint inhibitors in triple-negative breast cancer.Here DRG/Clarivate highlights the most impactful presentations on drug treatment landscapes. Over 2,100 oral presentations and posters were presented, showcasing exciting novel data across oncology, with the ultimate aim of improving cancer patient care. Darts-ip Design and Model Case Law DataĪs a result of the current COVID-19 pandemic, the annual ESMO Congress was held virtually between 19 th and 21 st September 2020.Transform the way you create, manage and protect intellectual property. Commercial Effectiveness & Launch Strategy.OFF-X Preclinical and Clinical Safety Intelligence.Portfolio Strategy & Business Development Advance innovation and accelerate patient outcomes.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
